
TS: I believe that device manufacturers have always considered extrusion as a process central to the development and fabrication of medical devices. With regard to many advanced devices this fact would be difficult to overstate! For this reason, many of the device manufacturers vertically integrate extrusion into the matrix of their manufacturing processes not just to possibly save money but to gain added insight into the relationship of the dynamics in the extruded part and how those dynamics affect the fabrication processes in manufacturing a medical device or catheter.
Here it is important to keep in mind that there is far more to an extruded tube than its dimensions. How a tube is extruded will affect most if not all of the manufacturing processes involved in fabricating devices. In the case of the catheter device pictured below, how the tube was extruded will affect the printing on both the catheter body and the extensions. The tipped distal end involves two lumens and two different materials, a clear polyurethane and two barium filled radiopaque stripes. The proximal end of this tube is over molded and bifurcated and connected two over molded extension tubes. For successful tipping and over molding, the dimensional attributes of the tube must be to specification to ensure smooth uniform tips. The criticality of the dimensional attributes involve more than just an inside diameter (ID) and an outside diameter (OD), but include the location of the stripes and the positioning of the lumen, which must be to specification for tipping and over-moulding to be successful.
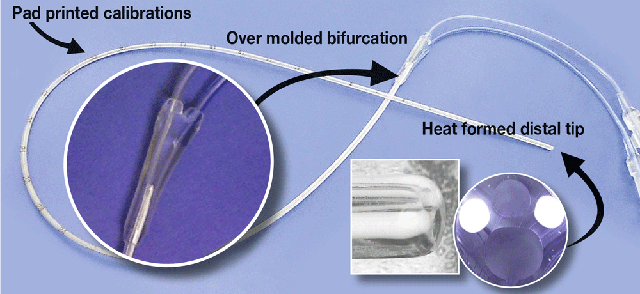
Thermal characteristics of the raw material is another important consideration when tipping and over-moulding and also should be considered if the part is to be ETO sterilised. With many elastomers, part dimensions can change when exposed to the heat the part is exposed to during sterilisation, or the heat it may see in a shipping container. This happens because when plastic is drawn down and cooled there is stress frozen into the plastic. To relieve this stress extruders often anneal parts. The part dimensions will change during annealing, but the latent stress in the tube is now gone and future dimensional changes from heat are eliminated.
In the design stage of an extrusion process for a new part it is useful to create a matrix of the desired attributes of form and function a particular part may be affected by the extruder and the process.
Process control in extrusion must be anticipated. This involves having a thorough and intuitive understanding of the interaction of the raw materials with the extruder and the extrusion tools. Monitoring and saving all process data is critical to developing and validating the extrusion process as it is with any manufacturing process
What materials does the process work best with and what are the advantages of the technology?
TS: I do not see that any particular material works best throughout the medical device industry. Although there are several families of materials that are used extensively throughout the device industry, I would have to say that the raw material selected for extrusion of a specific device depends on requirements of the device’s application. Physical characteristics generally considered first would be shore hardness, flex modulus, tensile strength, melting point, and service temperature. The matrix of material characteristics after that is specific to every application and what would work best in extrusion is largely based upon the competencies of the extruder. In short, what may work best for one extrusion company may not work best for another and to summarize this there is no best material. The use of custom formulated raw materials has been growing, along with the increase in highly specialised devices and advancements in antimicrobial compounds, advancements in drug delivery, and innovation throughout the medical industry.
How soon in the design process should the use of extrusion be considered and why?
Engineering design teams need to start considering extrusion requirements while brainstorming in the concept stage. Having the extrusion expert on the design team and participating in brainstorming during concept development will enhance concept development. This collaborative communication during the design phase helps the device maker more fully understand what is possible, but also gives the extruder a deeper understanding of the device maker’s expectations. The client and the extruder both benefit and the device gets to market faster.
What are challenges of the process and how can they be overcome?
TS: In the custom medical extrusion world there is not one formula the extruder follows to troubleshoot the extrusion challenges in producing the array of parts being demanded by today’s medical device industry. The devices continue to get smaller and smaller with precision becoming more and more critical. It is the same with functionality. Overcoming new challenges needs to be a collaborative process between the customer (device maker) and the extruder. Overcoming new challenges starts with good communication. Both parties need to have good understanding of the issues so that solutions are based upon intuitive decisions. Of course, both the device maker and the extruder have IP so a Mutual NDA is the first item in the development of a collaborative relationship based upon trust and the knowledge that both sides win by working together. Sharing knowledge is critical to success and both the extruder and the device maker need to be open with each other without giving away trade secrets.
How can extrusion experts such as MicroSpec share their knowledge with device designers at an early stage and then continue to manage customer expectations throughout the process?
TS: By participating in brainstorming during concept development of a new device we have experienced increased openness with our clients. With this our working relationship is strengthened which both sides in understanding the challenges we share in the project. As part of our extrusion service, we offer free technical advice when the client runs into technical problems and, if we do not have the answer, then we will refer to our own network of materials experts to find the answer and get back to the client.
Summary:
In the medical device industry, the extrusion process and the extrusion engineer are integral to the development of the ever increasing advanced medical devices. The extrusion process is innovating along with the device technology. Sharing knowledge of how extrusion affects device fabrication is critical to making intuitive decisions which will ultimately minimise the time to market for a new medical device.
Serving industry with quality stock and custom thermoplastic tubing, fittings and fabricated extruded products for over 30 years!
...more











